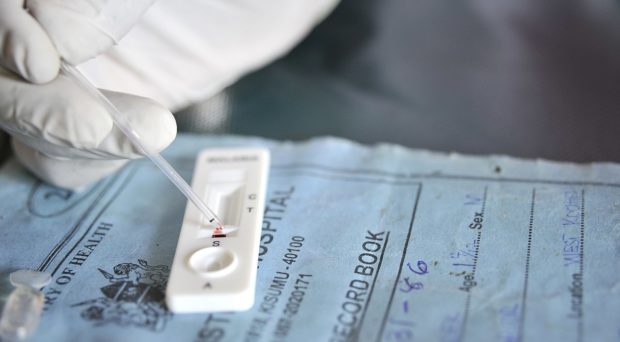
All diagnostics have some level of inherent subjectivity. Take a simple malaria rapid diagnostic test (RDT). The presence of a line on the immunochromatographic cassette indicates a positive result and therefore determines whether the patient should be given anti-malarials. What happens if the line is faint or your colleague can’t see it at all? Suddenly a binary output is blurred into a spectrum, and then either a second confirmatory test is required, or the user must make a judgement call.
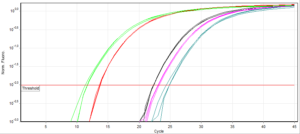
Quantitative tests such as ELISAs or qPCR that produce numerical values should be less vulnerable to interpretation, but still succumb. Using qPCR as an example, every assay has a unique fluorescence threshold. This can vary with the model of PCR machine and the level of background fluorescence in the mastermix/probes you use. It is generally accepted as a variable that needs to be determined for each assay. The cycle number at which any given sample reaches the predetermined level of fluorescence is called the cycle threshold (Ct). To be considered positive, this Ct must fall before a limit termed the ‘Ct cut-off’.
There are two schools of thought on defining Ct cut-offs. Firstly, any sample that crosses the fluorescence threshold within the completed run time (normally ~40 cycles), should be defined ‘positive’ i.e. there is no imposed cut-off. This simply assumes that amplification would not be observed without target DNA. Secondly, that Ct cut-offs should be set according to the limit of detection (LOD)- discovered through testing dilutions of control DNA and determining when positive results are no longer observed. This practice acknowledges and compensates for the inherent risk of trace contamination between samples, or non-specific amplification. However, this can lead to low level infections being ruled negative.
Aside from the obvious clinical implications of undiagnosed malaria infections, these patients contribute to the overall infectious reservoir of malaria parasites. This threatens the success of malaria elimination programs, where low-parasitaemic infections prevail.

The non-standardised approach to setting diagnostic thresholds- particularly ‘in-house’ tests- can have a significant impact on the interpretation of data. Furthermore, it makes the compilation of diagnostic data for meta-analyses incredibly difficult. In attempt to account for this, several statistical methods have been developed, including receiver operating characteristic (ROC) curves and bivariate analyses, as well as a quality assessment framework ‘QUADAS’ to compare diagnostic accuracy studies. As the WHO work towards building an Essential Diagnostics Lists (proposed earlier this year), it will become increasingly important to provide full disclosure of the thresholds used alongside primary data for the most effective analysis.

Comments